|
Two patients with IMT were enrolled already in the dose escalation phase: for one of these, a rapid and sustained partial response was seen.
 Clinical proof of concept, culminating in the recent approval by the Food and Drug Administration of the Pfizer drug crizotinib followed in record time.The drug was approved together with a companion diagnostic for detection of patients eligible for therapy.This remarkable example of the coming of age of personalized medicine in cancer therapy is hopefully only an auspice of things to come in a rapidly developing field. Clinical proof of concept, culminating in the recent approval by the Food and Drug Administration of the Pfizer drug crizotinib followed in record time.The drug was approved together with a companion diagnostic for detection of patients eligible for therapy.This remarkable example of the coming of age of personalized medicine in cancer therapy is hopefully only an auspice of things to come in a rapidly developing field.
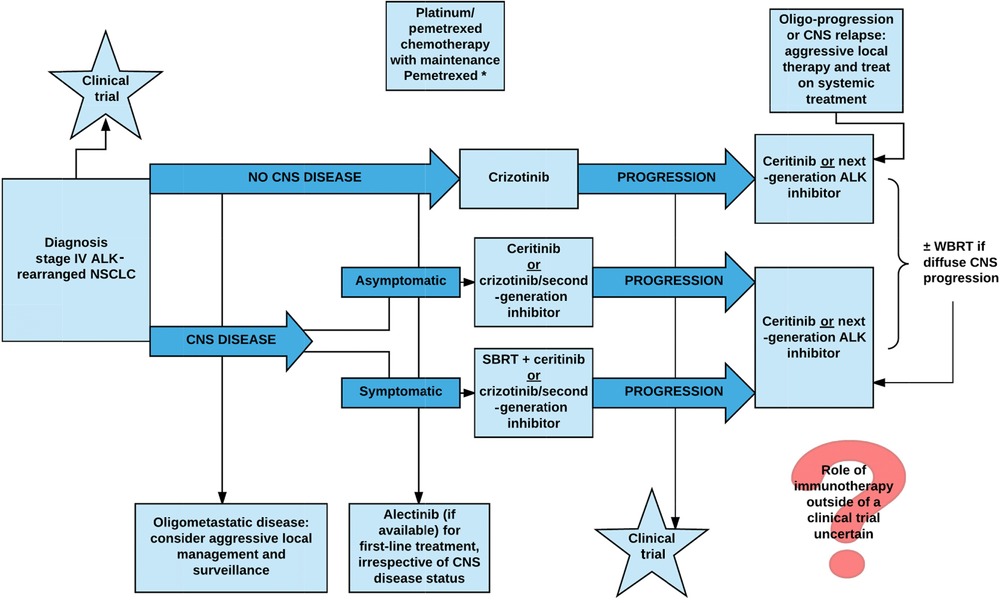
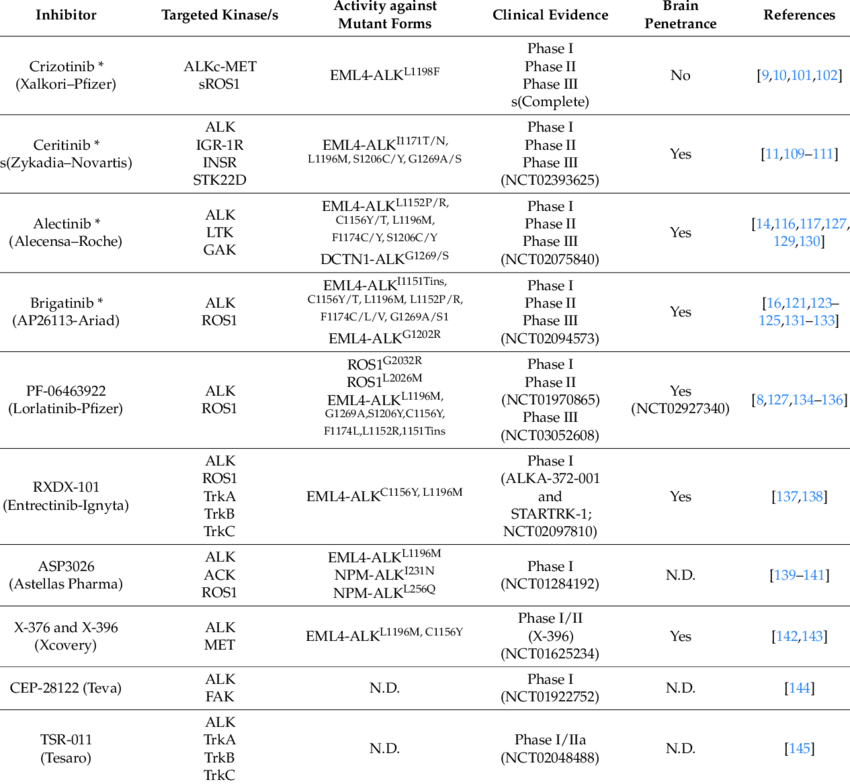
It is also emerging that acquired resistance to crizotinib may additionally occur through ALK-independent mechanisms, which still need to be elucidated in detail. Here we discuss the factors that led to such a rapid approval of a targeted agent, and we describe the second-generation compounds currently in development. As with NPMALK, there is much convincing preclinical evidence in support of the oncogenic nature of EML4ALK, the requirement for ALK kinase activity in maintenance of EML4ALK-dependent tumor cell growth and of the capacity of selective small molecule kinase inhibitors of ALK to induce cell death in such tumors ( Christensen et al., 2007; Galkin et al., 2007; Choi et al., 2008; Koivunen et al., 2008; McDermott et al., 2008; Soda et al., 2008 ). Subsequent studies of tissue samples from NSCLC patients aimed at further characterizing ALK-positive NSCLC have led to the identification of a relatively well defined potential patient population, characterized by specific clinicalpathological features. Studies with c-Met kinase revealed that crizotinib has a classical ATP-competitive mechanism of action and as is often the case for such inhibitors, it was subsequently found to cross-react with a few off-target kinases. In particular, potent activity of the drug on ALK was revealed through selectivity profiling in biochemical assay and ALK-driven cellular models ( Christensen et al., 2007 ). Alk Inhibitor Sequence Trial Of CrizotinibA multi-indication Phase I clinical trial of crizotinib in solid tumors and lymphomas had already been initiated, with the drug described as a c-MetHepatocyte Growth Factor tyrosine kinase inhibitor (ClinicalTrials.gov Identifier: NCT00585195), when identification of the genetic rearrangement involving ALK in NSCLC was first reported ( Soda et al., 2007 ). In 2008, while preclinical data supporting a therapeutic rationale for targeting ALK in NSCLC was still emerging, ALK-positive patients started to be enrolled in this already ongoing Phase I trial. Alk Inhibitor Sequence Registration Of TheALK cross-reactivity of crizotinib, apparently initially seen as a possible path for registration of the compound in niche indications such as chemotherapy resistant ALCL, now became a major opportunity. Thus, patient screening and enrollment of ALK-positive subjects into the trial was initiated, using a methodology based on the break apart probe FISH (fluorescent in situ hybridization) technique, with a kit specifically developed for detecting ALK translocation in patient tumor samples ( Perner et al., 2008 ). Within a few months, impressive preliminary data on clinical response in these patients became available. A dedicated Phase III clinical trial focused on ALK-positive NSCLC patients was completed in 2010 ( Kwak et al., 2010 ), barely 3 years after the first description of this genetic lesion. After the standard dose escalation Phase I that defined the recommended dose of 250 mg twice a day per 28-day cycle, an expanded cohort of ALK-positive NSCLC was selected for treatment. Approximately 1500 NSCLC patients were screened by FISH, identifying 82 patients considered eligible and then enrolled in the expanded cohort study. Most of these patients had received previous therapy and almost half were heavily pre-treated. The overall objective response rate in this study was 57 (47 out of 82 patients, with 46 confirmed partial response and 1 complete response), with a further 33 of patients (27 out of 82) in stable disease. Alk Inhibitor Sequence Free Survival WasThe estimated probability of 6-month progression-free survival was 72. To date, the median overall survival time from initiation of crizotinib has not been determined, but 1-year overall survival was 74 and 2-year overall survival was 54 ( Kwak et al., 2010; Shaw et al., 2011 ). The most frequently reported were gastrointestinal toxicities, with grade 1 nausea and diarrhea and visual disturbances, but with no abnormalities detected in ophthalmological examination. Increased levels of hepatic transaminases were also observed, but only reaching grade 3 in a limited number of patients (5 and 6 for ALT and AST, respectively). Two randomized Phase III clinical trials in ALK-positive NSCLC are currently underway to compare the activity of crizotinib to standard of care. Nevertheless, based on the impressive responses observed in Phase III trial, the Food and Drug Administration (FDA) approved crizotinib for treatment of ALK rearranged NSCLC, under its accelerated approval program, on August 26, 2011. For at least two patients with ALK-positive ALCL treated at the recommended Phase II dose, signs of clinical benefit were seen within a remarkably short treatment period, with a PR and a CR achieved ( Gambacorti-Passerini and Messa, 2011 ).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
